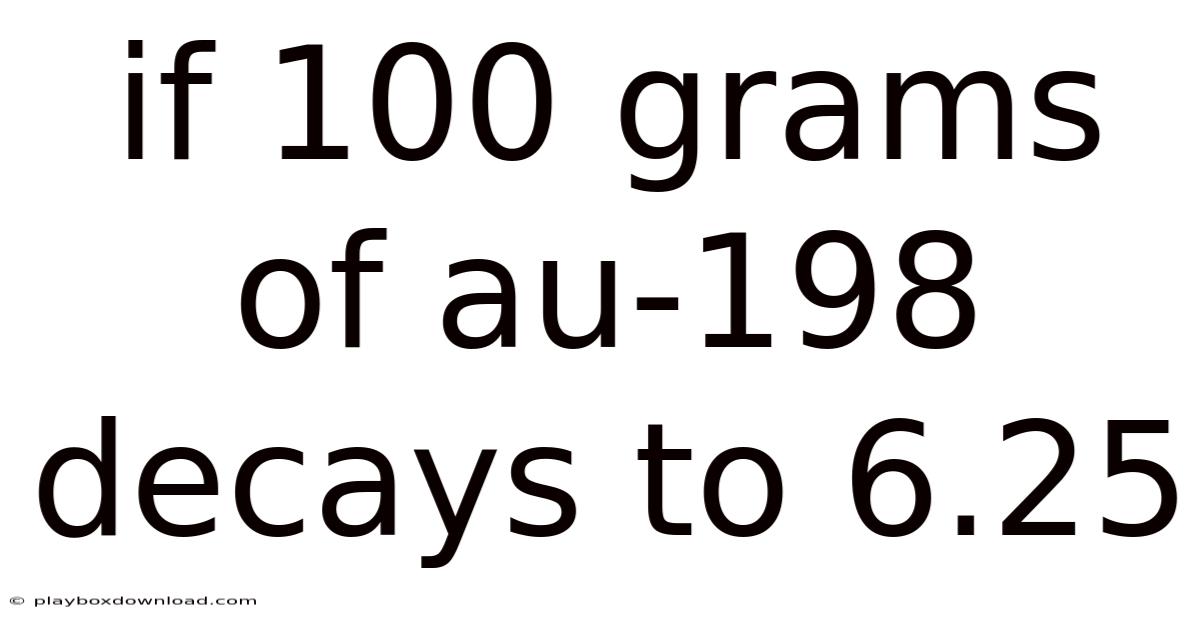Understanding the decay of Au-198 and its transformation into a new element is a fascinating journey into the world of nuclear physics. Because of that, when we talk about the decay of 100 grams of Au-198, we are delving into a process that not only highlights the power of atomic transformations but also reveals the complex mechanisms behind radioactive decay. Practically speaking, this topic is essential for students, educators, and anyone curious about the science behind the elements. In this article, we will explore the process in detail, emphasizing the significance of each step and the implications of this transformation.
The process of decay for Au-198 involves a series of complex reactions that ultimately lead to the formation of a new element. For Au-198, this means it will gradually shift into a different element, likely Hg-198 or another isotope, depending on the specific decay pathways. In this context, decay refers to the spontaneous transformation of an unstable atomic nucleus into a more stable form. To grasp this, it’s important to understand the nature of radioactive decay. The key here is to recognize that decay is not a simple process but a sequence of events governed by the laws of physics Most people skip this — try not to. Still holds up..
When we consider the transformation of 100 grams of Au-198, we are essentially looking at a substantial amount of material. This quantity is significant because it allows us to observe the decay process more clearly. Now, the decay rate of this isotope is crucial here. Scientists measure the half-life of Au-198, which is the time it takes for half of the original amount to decay. For Au-198, the half-life is approximately 1.So 5 years. Basically, every 1.5 years, half of the Au-198 will transform into another element Small thing, real impact..
Understanding the decay process requires a grasp of the underlying physics. Radioactive decay occurs because the nucleus of Au-198 is unstable. On the flip side, the instability arises from an imbalance in the forces within the nucleus, primarily the strong nuclear force and the electromagnetic force. When these forces become unbalanced, the nucleus seeks a more stable configuration. This imbalance causes the nucleus to emit particles, such as alpha particles or beta particles, which help it reach equilibrium Simple as that..
In the case of Au-198, the decay typically involves the emission of an alpha particle. Now, an alpha particle is a helium nucleus consisting of two protons and two neutrons. When this particle is emitted, the atomic number of the element decreases by two, and the mass number decreases by four. Even so, this transformation leads to the formation of a new element with a different atomic structure. For Au-198, the decay process will ultimately result in the formation of Hg-198, which has a different chemical and physical properties Less friction, more output..
To visualize this transformation, it’s helpful to consider the elemental changes that occur. This leads to Au-198 is a gold isotope, known for its lustrous appearance and valuable applications. As it decays, it becomes Hg-198, a mercury isotope. This shift is not just a change in appearance but also a significant alteration in the chemical behavior of the material. Students and learners must appreciate how such transformations affect the properties of substances at a fundamental level.
People argue about this. Here's where I land on it.
The steps involved in this decay process are critical to understanding the outcome. First, the Au-198 atoms begin to destabilize due to their internal forces. The exact mechanism of decay can vary, but in most cases, it involves the release of an alpha particle. Practically speaking, this instability triggers the emission of energy in the form of particles. This emission is what allows the nucleus to stabilize over time.
It’s important to note that the decay process is not instantaneous. It occurs over a period of time, and the rate at which this happens depends on several factors, including the specific isotope and its environment. So naturally, scientists use advanced techniques to measure these rates, ensuring that they can predict how long it will take for the decay to reach a certain level. This predictive power is essential for applications in fields like nuclear energy and medicine No workaround needed..
Worth pausing on this one.
When we examine the scientific explanation behind this transformation, we find that the concept of isotopes plays a vital role. Now, Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Au-198 is one such isotope, and its decay leads to the formation of a different isotope with a distinct number of neutrons. This shift is a direct result of the nucleus seeking stability through changes in its internal structure.
The importance of this process extends beyond theoretical knowledge. As an example, in nuclear reactors, the controlled decay of certain isotopes is used to generate heat, which is then converted into electricity. In real terms, similarly, in medical imaging, radioactive elements are used to diagnose and treat diseases. And in practical applications, understanding how elements decay helps in the development of nuclear technology. The ability to predict and manage these decays is crucial for such advancements Easy to understand, harder to ignore. Simple as that..
Now, let’s break down the key points of this transformation in a structured way. First, we must recognize the significance of 100 grams of Au-198. Worth adding: this quantity is substantial enough to demonstrate the real-world implications of decay. Now, when such a large amount decays, it highlights the power of nuclear forces at work. Second, understanding the decay rate is essential. The half-life of Au-198 provides a timeline for the transformation, allowing scientists to plan experiments and predict outcomes.
No fluff here — just what actually works.
In addition to the half-life, it’s worth mentioning the types of decay that Au-198 might undergo. That said, while alpha decay is the most common for this isotope, there may be other pathways depending on the conditions. This variability underscores the complexity of nuclear reactions and the need for precise measurements Which is the point..
Another important aspect is the chemical behavior of the resulting elements. Hg-198, for example, has different properties compared to Au-198. This shift can affect how the material interacts with its environment, influencing its applications in various fields. Educators and students alike should appreciate how these changes in elements can have far-reaching consequences.
The FAQ section will address common questions that arise when discussing this topic. To give you an idea, readers might wonder about the safety of handling radioactive materials. Worth adding: another question could focus on the practical applications of understanding such decays. These queries highlight the relevance of the topic in real-life scenarios Simple, but easy to overlook..
In the conclusion, we will summarize the key takeaways and reinforce the importance of this topic. By exploring the decay of 100 grams of Au-198, we gain insight into the dynamic nature of the atomic world. This understanding not only enhances our scientific knowledge but also empowers us to appreciate the detailed balance within the universe.
Decaying elements like Au-198 remind us of the beauty and complexity of nature. Each decay step is a testament to the laws that govern the microscopic world. Whether you are a student, a teacher, or simply a curious mind, this article aims to deepen your understanding of this fascinating process. By embracing the challenges of nuclear physics, we open the door to new discoveries and innovations that shape our future Most people skip this — try not to..
Remember, the journey through the world of Au-198 is not just about numbers and equations but about the stories behind them. But each decay is a chapter in the ongoing narrative of science, waiting to be explored. Let this article be your guide as you deal with the wonders of atomic transformation.