A Hydrate Of Cocl2 With A Mass Of 6.00 G
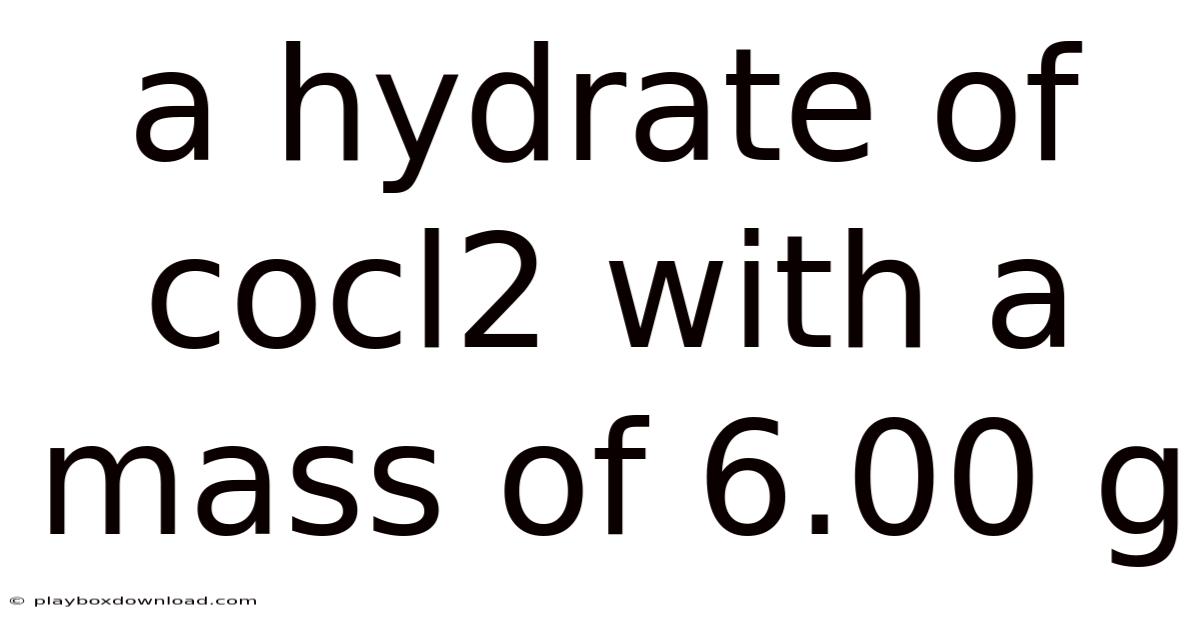
Understanding the hydrate of cocl2 with a mass of 6.00 g is a fascinating journey into the world of inorganic chemistry. This compound, known for its unique structure and properties, plays a significant role in various scientific and industrial applications. In this article, we will delve into the details of this hydrate, exploring its composition, characteristics, and importance.
When we talk about the hydrate of cocl2, we are referring to a crystalline solid that contains a specific number of water molecules associated with its crystal structure. The term "hydrate" indicates the presence of water molecules in the lattice of the compound. In this case, the mass of the hydrate is carefully measured at 6.00 grams, which is crucial for understanding its physical and chemical properties.
The importance of this hydrate lies in its stability and the way it interacts with other substances. The specific mass of the hydrate is essential for identifying it accurately in laboratory settings. It helps chemists determine the purity and composition of the compound, ensuring that it meets the required standards for its intended use.
To begin with, let’s clarify what cocl2 is. Cocl2 is a complex ion composed of cobalt and two chloride ions. When it forms a hydrate, it incorporates water molecules into its crystal structure. The mass of this hydrate, 6.00 g, is a critical piece of information that scientists use to analyze its behavior in various environments. Understanding this mass helps researchers predict how the hydrate will react under different conditions, such as temperature changes or when exposed to moisture.
Now, let’s explore the scientific explanation behind the formation of this hydrate. The process involves the coordination of water molecules around the cobalt ions. These water molecules stabilize the structure of the hydrate, making it more robust and resistant to degradation. The chemical bonds formed between the cobalt ions and the water molecules are vital for maintaining the integrity of the hydrate. This stability is what makes the hydrate of cocl2 so interesting for both academic and industrial purposes.
In practical applications, the hydrate of cocl2 finds its way into various fields. For instance, it can be used in analytical chemistry for the development of sensors and detectors. The presence of water molecules in the hydrate can influence the sensor's sensitivity and accuracy. Additionally, this compound is relevant in the pharmaceutical industry, where it may be used in formulations that require precise control over moisture content.
When we discuss the FAQ section, readers might have questions about the properties of this hydrate. For example, how does the mass affect its solubility? The answer lies in the balance between the hydrate and its surrounding environment. A higher mass can influence the solubility, making it more or less soluble depending on the conditions. Understanding this relationship is crucial for optimizing its use in different applications.
Moreover, the conclusion of this article highlights the significance of the hydrate of cocl2 in research and industry. Its unique characteristics make it a subject of interest for chemists and scientists aiming to develop new materials or improve existing processes. The importance of this hydrate extends beyond its chemical properties; it also represents a bridge between theoretical knowledge and practical application.
In summary, the hydrate of cocl2 with a mass of 6.00 g is more than just a numerical value; it is a key element in the field of chemistry. By understanding its structure, properties, and applications, we can appreciate the intricate world of inorganic compounds. This article has aimed to provide a comprehensive overview, encouraging readers to explore further into the fascinating realm of chemistry. Remember, every detail matters in the pursuit of knowledge, and this hydrate serves as a prime example of that principle.
Through this exploration, we not only learn about the hydrate of cocl2 but also gain insights into the broader implications of its study. Whether you are a student, educator, or professional, this article offers valuable information that can enhance your understanding of this intriguing compound. Embrace the challenge of learning about such topics, and let your curiosity drive you to uncover more about the wonders of science.
Continuing from theprovided text:
The study of the hydrate of CoCl₂, particularly at the precise mass of 6.00 g, offers a compelling case study in the intricate interplay between structure and function in inorganic chemistry. This specific hydrate composition, often denoted as CoCl₂·6H₂O, is not merely a stoichiometric curiosity but a tangible example of how molecular architecture dictates material behavior. The water molecules, bound through coordination bonds to the cobalt ions, are far more than passive spectators; they actively sculpt the compound's physical and chemical identity, influencing properties like color, solubility, thermal stability, and reactivity.
Understanding the precise mass of 6.00 g is crucial for several reasons. It allows for accurate stoichiometric calculations in synthesis and analysis, ensuring reproducibility in experiments. It provides a benchmark for characterizing the hydrate's purity and composition. Furthermore, this mass serves as a reference point for investigating how variations in hydration state (e.g., CoCl₂·2H₂O vs. CoCl₂·6H₂O) alter the compound's properties, offering insights into the thermodynamics of hydrate formation and the factors governing water affinity.
The significance of this hydrate extends beyond the laboratory bench. Its well-documented color change from deep blue (anhydrous) to pink (hexahydrate) upon hydration has made it a popular visual indicator in educational demonstrations and practical applications, such as humidity sensors. The stability conferred by the cobalt-water bonds, as initially highlighted, underpins its reliability in these roles. Moreover, the principles governing its formation and stability are mirrored in countless other metal hydrates and coordination polymers, making CoCl₂·6H₂O a fundamental model system.
In essence, the hydrate of cobalt(II) chloride, especially at the 6.00 g mass, embodies the profound connection between molecular structure and macroscopic properties. It is a testament to the elegance of coordination chemistry, where simple ions and water molecules assemble into complex, functional entities. Its study provides not only practical tools for analysis and sensing but also deepens our fundamental understanding of how matter organizes itself at the molecular level. This compound, therefore, stands as a significant milestone in the exploration of inorganic chemistry, bridging theoretical concepts with tangible, observable phenomena and demonstrating the enduring value of meticulous chemical investigation.
Conclusion:
The hydrate of cobalt(II) chloride, precisely quantified at 6.00 g, is far more than a simple salt compound. It is a sophisticated assembly where cobalt ions and water molecules engage in vital coordination bonds, creating a structure of remarkable stability that underpins its diverse applications. From its role in sensitive analytical sensors and moisture-sensitive pharmaceutical formulations to its use as a vivid educational tool and humidity indicator, this hydrate exemplifies the practical importance of understanding molecular interactions. Its well-defined mass serves as a critical parameter for research, synthesis, and characterization. Ultimately, CoCl₂·6H₂O represents a cornerstone in inorganic chemistry, illustrating how fundamental principles of bonding and structure translate into real-world functionality and scientific insight. Its study continues to inspire both practical innovation and theoretical advancement, solidifying its place as a fascinating and indispensable subject within the chemical sciences.
Continuingfrom the established foundation of cobalt(II) chloride hexahydrate's significance, we must acknowledge its profound impact on both fundamental science and applied technology. Its unique behavior as a humidity indicator transcends mere curiosity; it represents a sophisticated interplay between chemical structure and environmental responsiveness. The reversible hydration/dehydration cycle, governed by the delicate balance of cobalt(II) ion hydration and chloride counterion interactions, exemplifies a dynamic equilibrium critical to sensor functionality. This principle extends beyond CoCl₂·6H₂O, informing the design of advanced materials like metal-organic frameworks (MOFs) and zeolites, where precise control over water uptake and release is paramount for gas storage, separation, and catalysis.
Furthermore, the compound's role as an educational cornerstone cannot be overstated. Its vivid color transformation provides an unparalleled, tangible demonstration of coordination chemistry concepts – ligand field theory, crystal field stabilization energy, and the profound influence of hydration on electronic transitions. This visual accessibility bridges the gap between abstract theory and observable reality, fostering deeper student engagement and comprehension. The precise 6.00 g mass specification mentioned earlier is not merely a numerical value; it serves as a critical parameter in laboratory protocols, ensuring reproducibility in experiments exploring hydrate stoichiometry, thermal decomposition pathways, and kinetic studies of dehydration.
The investigation of CoCl₂·6H₂O also illuminates broader thermodynamic principles. The enthalpy and entropy changes associated with its hydration process offer valuable insights into the strength and nature of metal-ligand bonds compared to other cations. This understanding contributes to predictive models for hydrate stability across diverse systems, aiding in the prediction of phase behavior in geological repositories, industrial processes involving gas hydrates, and the formulation of stable pharmaceuticals requiring controlled moisture environments. Its study thus exemplifies how a single, well-characterized compound can serve as a microcosm, revealing universal laws governing molecular assembly and interaction.
Ultimately, cobalt(II) chloride hexahydrate stands as a testament to the elegance and utility inherent in inorganic chemistry. It bridges the gap between fundamental molecular interactions and tangible, observable phenomena. Its study provides not only practical tools for analysis and sensing but also deepens our fundamental understanding of how matter organizes itself at the molecular level. This compound, therefore, represents a significant milestone in the exploration of inorganic chemistry, demonstrating the enduring value of meticulous chemical investigation and the profound connection between molecular structure and macroscopic properties. Its legacy continues to inspire both practical innovation and theoretical advancement, solidifying its place
as a foundational element in the ongoing evolution of chemistry and materials science.
In conclusion, the study of CoCl₂·6H₂O offers a multifaceted and rewarding journey into the heart of chemical principles. From its practical applications in material science and analytical chemistry to its invaluable role in education and thermodynamic understanding, this seemingly simple compound reveals a wealth of complexity and interconnectedness. Its enduring relevance demonstrates the power of careful observation, precise experimentation, and theoretical modeling in unlocking the secrets of the molecular world. The insights gained from investigating CoCl₂·6H₂O continue to shape research across diverse fields, ensuring its place as a cornerstone of chemical knowledge for generations to come.
Latest Posts
Latest Posts
-
Kumon Math Level J Answer Book
Mar 24, 2026
-
Till We Have Faces Summary Chapter 1
Mar 24, 2026
-
Who Initiates Chain Of Custody For Items Collected
Mar 24, 2026
-
Fill In The Blank In The Triangle Below Y
Mar 24, 2026
-
What Is Required To Access Dynamic Study Modules
Mar 24, 2026